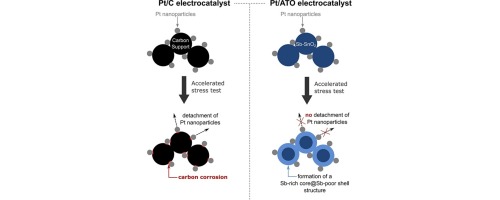
Paper by David Riassetto 2017
The paper "Benefits and limitations of Pt nanoparticles supported on highlyporous antimony-doped tin dioxide aerogel as alternative cathodematerial for proton-exchange membrane fuel cells" has been published in Applied Catalysis B: Environmental