OBJECTIVES
The stability of therapeutic proteins at interfaces is a pivotal topic in the pharmaceutical industry where biotherapeutics are produced and formulated and in the device industry where reconstitution and injection systems are designed for the administration of the drugs to the patient. Our collaborative partners from these industrial sectors are :
3 research topics are currently developed:
3 research topics are currently developed:
- The effect of dehydration on proteins adsorbed at material surfaces
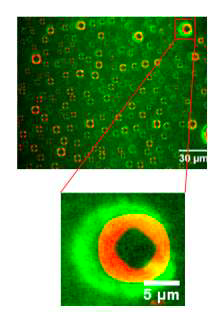
Proteins tend to adsorb easily at surfaces and, in the presence of air, can become partially dehydrated, when the protein solution is agitated. We develop experimental protocols to control the exposure to air and analyse the effect of a dynamic material-liquid-air interface on protein stability.
- The interfacial activity of proteins at high concentration
Therapeutic proteins are formulated at high concentrations, at which protein-protein interactions accompany interfacial phenomena. We therefore are interested in analysing the impact of the cooperative protein behaviour at interfaces and how protein-protein interactions influence their adsorption/desorption kinetics.
- The role of surfactants at interfaces
Surfactants are commonly known to stabilize therapeutic protein solutions by kinetic competition at the interface. We analyse their stabilisation efficiency as a function of the material surface and in the presence of air.
Researchers
Group Members
Highlights
The observation, in real time, by fluorescence and interference microscopies of a dynamic triple interface, have demonstrated that human insulin adsorbed on a hydrophobic surface aggregates into amyloid fibres due to repeated dehydration. This work has direct implications on established handling protocols for formulations containing therapeutic proteins in order to guarantee their stability. This topic is also at the heart of the LMGP-EVEON LabCom.