OBJECTIFS
La stabilité des protéines thérapeutiques aux interfaces est un sujet essentiel dans l’industrie pharmaceutique où les bio-médicaments sont produits et formulés, ainsi que dans l’industrie des dispositifs où des systèmes de reconstitution et d’injection sont conçus pour l’administration des médicaments au patient. Nos partenaires collaboratifs de ces secteurs industriels sont:
3 thèmes de recherche sont actuellement développés:
3 thèmes de recherche sont actuellement développés:
- L’effet de la déshydratation sur les protéines adsorbées sur les surfaces des matériaux
Les protéines ont tendance à s'adsorber facilement sur les surfaces et, en présence d'air, peuvent devenir partiellement déshydratées lorsque la solution de protéines est agitée. Nous développons des protocoles expérimentaux pour contrôler l'exposition à l'air et analysons l'effet d'une interface dynamique matériau-liquide-air sur la stabilité des protéines.
- L'activité aux interfaces des protéines à haute concentration
Les protéines thérapeutiques sont formulées à des concentrations élevées, auxquelles des interactions protéine-protéine accompagnent les phénomènes aux interfaces. Nous sommes donc intéressés par l'analyse de l'impact du comportement coopératif des protéines aux interfaces et par la manière dont les interactions protéine-protéine influencent leur cinétique d'adsorption / désorption.
- Le rôle des tensioactifs aux interfaces
Les tensioactifs sont généralement connus pour stabiliser les solutions de protéines thérapeutiques par compétition cinétique à l'interface. Nous analysons leur efficacité de stabilisation en fonction de la surface du matériau et en présence d’air.
Chercheurs
Membres
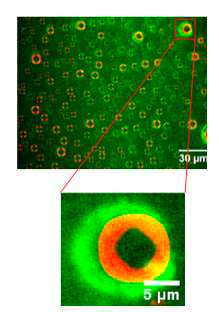
FAIT MARQUANT
L'observation, en temps réel, par microscopie de fluorescence et d'interférence d'une triple interface dynamique, a montré que l'insuline humaine adsorbée sur une surface hydrophobe s'agrège en fibres amyloïdes en raison d'une déshydratation répétée. Ces travaux ont des implications directes sur les protocoles de manipulation établis pour les formulations contenant des protéines thérapeutiques. Ces résultats ont mené à l’établissement d’un laboratoire commun entre EVEON et le LMGP dont le but est l’optimisation des systèmes de préparation et d'administration automatisées des médicaments afin de garantir leur stabilité.