Publication de Quentin Lubart 2017
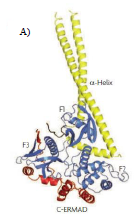
Le papier "Role of Phosphorylation in Moesin Interactions with PIP2-Containing Biomimetic Membranes" a été publié dans Biophysical Journal
Ici vous trouverez les papier de Quentin Lubart